  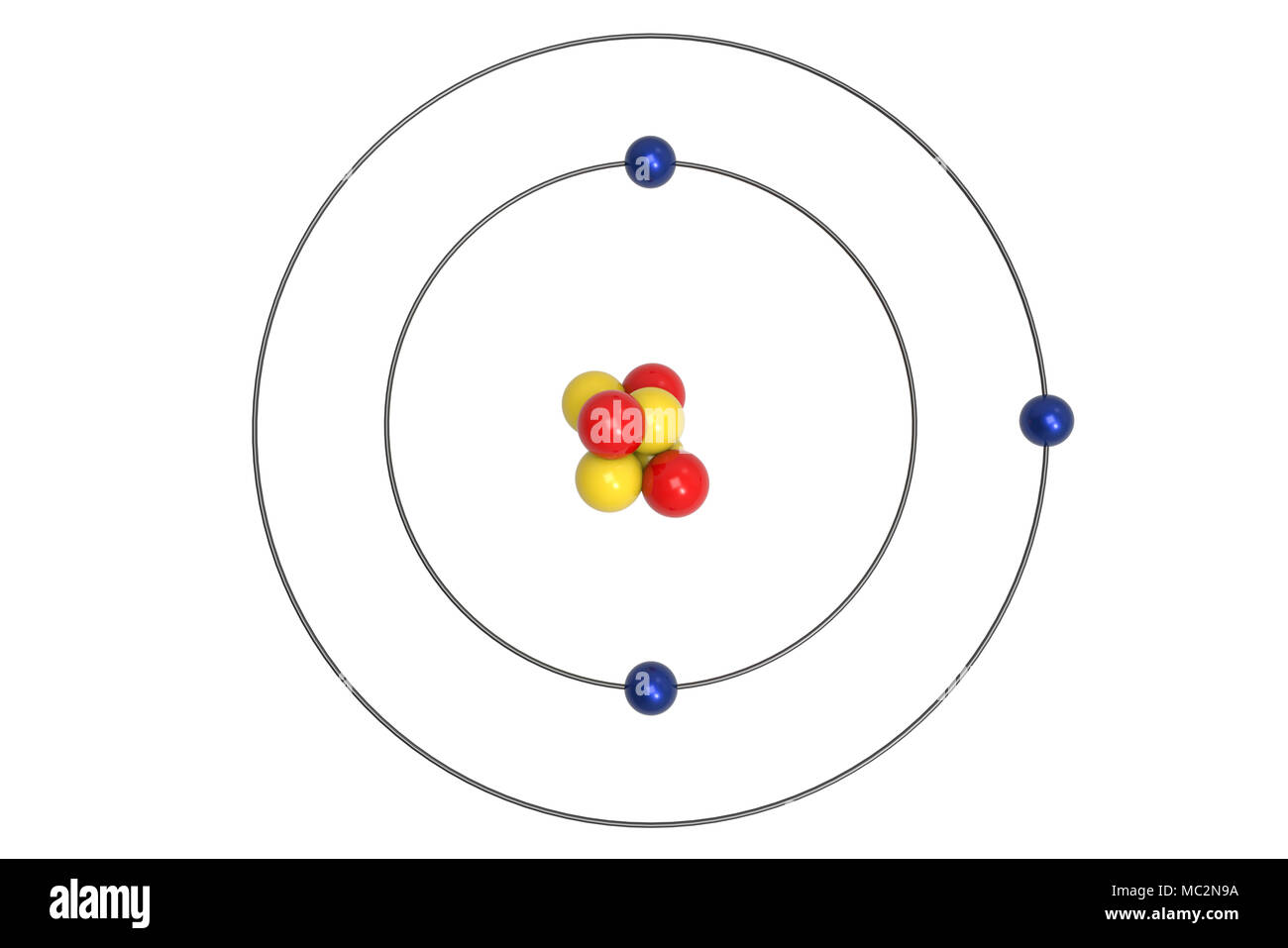
Number of electrons = 3 = Atomic number of Lithium Now, we will calculate the number of electrons for the Lithium atom.Īctually, the number of electrons, as well as protons in an atom, is the same, as they both are equal to the atomic number of the atom. In the above diagram, p + = protons & n° = neutrons. Using these values we will draw the nucleus of the Lithium atom. Hence, number of neutrons in the Lithium atom = 4 Number of neutrons in the Lithium atom = 7 – 3 = 4 Now, finding the number of neutrons by applying the formula: Looking at the Lithium box that we drew above, we can say that the atomic mass of the Lithium atom is 6.938.Īfter rounding it up to the nearest whole number we get 7.Īlso, as calculated above, the number of protons in the Lithium atom is 3. Number of neutrons = Atomic mass (rounding it up to the nearest whole number) – Number of protons The following formula can be used for calculating the number of neutrons in an atom Therefore, for the Lithium atom, the number of protons = atomic number = 3įurther, we will calculate the number of neutrons present in the Lithium atom. In the case of the Lithium atom, the atomic number is 3. The number of protons for any atom is always equal to the atomic number of that atom. Let us begin by drawing the nucleus for the Lithium atom, for which we will have to calculate the number of protons and neutrons in the Lithium atom.įirst, we will calculate the number of protons Now, we can draw the Bohr model of the Lithium atom with the help of this information. Lithium has electronic configuration 2s 1.The information that we can derive from the above-mentioned Lithium box is as follows: Lithium belongs to group 1 and period 2 of the periodic table. The Lithium atom consists of 3 protons and 4 neutrons in the nucleus, and 3 electrons revolve around the Lithium atom in 2 shells i.e. The electron that absorbs energy jumps upwards while the electron that loses energy jumps downward 
The electrons are also capable of jumping up and down from their orbits into the higher or lower orbit, respectively. Similarly, the electrons housed in the shell farthest from the nucleus (here, the name of the shell may differ with the type of atom) have the highest energy and are called, valence electrons, while the shell is known as the valence shell the K shell are said to be in the ground state, similar to their assigned shell they also carry maximum energy out of all the electrons. The electrons located in the shell nearest to the nucleus i.e. The electron carrying capacity of the shell also increases outwards The innermost shell is assigned the lowest value which increases outwards i.e. The shells are also given names in numerological as well as alphabetical order. Usually, the smallest shell, located nearest to the atom has the least energy while the largest and farthest shell has maximum energy. These are pre-determined paths and have fixed energies associated with them due to which they are also known as energy levels. Shells: The electrons move around the atoms in their orbits, known as shells.They are not stagnant like other atomic particles and circulate around the nucleus. Electrons: The only type of atomic particles present outside the nucleus in the atomic space.Neutrons: These are also located inside the nucleus but are charge-neutral species.Protons: These are located inside the nucleus and confer a positive charge to it.The other atomic particles present in the nucleus are neutrons. 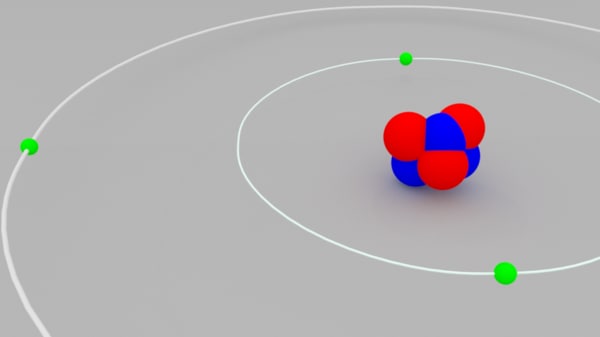
It exhibits a positive charge owing to its constituent particles i.e.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
